- Distinguish short-range and long-range order in solids using real-space correlations.
- Classify solids as crystalline, polycrystalline, or amorphous, and explain why this matters for thermal physics.
- Write down the full many-particle Hamiltonian of a solid and identify the origin of its complexity.
- Explain, at a roadmap level, how bonding, lattice stiffness, vibrations, and phonons connect to thermal observables.
- Identify which later approximations are needed to reach heat capacity, thermal expansion, and thermal conductivity.
1.1 Definitions
| Symbol | Meaning |
|---|---|
| \(\mathbf R_i\) | position of nucleus / ion \(i\) |
| \(\mathbf r_j\) | position of electron \(j\) |
| \(\rho(\mathbf r)\) | one-particle density |
| \(P(\mathbf r_1,\mathbf r_2)\) | two-particle density |
| \(g(\mathbf r)\) | pair-distribution function (or radial distribution function in isotropic systems) |
| \(S(\mathbf K)\) | structure factor |
| \(\mathbf K\) | Fourier mode |
| \(H\) | many-particle Hamiltonian |
| \(C_V\), \(\alpha\), \(\kappa\) | heat capacity, thermal expansion coefficient, thermal conductivity |
1.2 Why Begin a Course on Thermal Properties with Structure?
A first description of thermal, mechanical, electric, or magnetic behavior can often be given at a phenomenological level, using thermodynamics or continuum theories. However, a deeper understanding requires an atomic picture: solids are many-particle quantum systems made of interacting nuclei and electrons. This is the microscopic starting point of solid-state physics.
For thermal physics, this point is especially important. A solid is not merely a rigid arrangement of atoms. At finite temperature, the atoms fluctuate about their equilibrium positions, and these collective motions dominate much of the thermal behavior that we measure macroscopically. The central question of the course is therefore:
Which microscopic degrees of freedom carry thermal energy in a solid, and how do they produce measurable quantities such as \(C_V\), \(\alpha\), and \(\kappa\)?
This lecture sets up the language needed to answer that question.
1.3 Short-Range and Long-Range Order
1.3.1 Positional Order as a Correlation Problem
In condensed matter, atomic positions are usually not completely random. Interatomic forces produce correlations, and these correlations may persist either only locally or over arbitrarily large distances.
A convenient formal language is provided by distribution functions. The one-particle density is
and the two-particle density is
The corresponding pair-correlation function is
These definitions are the standard statistical characterization of structure in disordered matter.
For a homogeneous system, \(\rho(\mathbf r)=\rho\) is constant, and \(g\) depends only on the difference of its arguments: \[ g(\mathbf r_1,\mathbf r_2)=g(\mathbf r_1-\mathbf r_2). \] If the system is also isotropic, then \(g\) depends only on \(r=|\mathbf r|\), so one writes simply \(g(r)\) which is also often called radial distribution function.
1.3.2 Short-Range versus Long-Range Order
The key distinction is encoded in the large-distance behavior of correlations.
If no long-range order is present, then distant atomic positions become uncorrelated: \[ P(\mathbf r_1,\mathbf r_2)\to \rho(\mathbf r_1)\rho(\mathbf r_2) \qquad \text{for } |\mathbf r_1-\mathbf r_2|\to\infty. \] Equivalently, \[ g(r)\to 1 \qquad \text{for } r\to\infty. \]
This motivates the total correlation function \[ h(\mathbf r)=g(\mathbf r)-1, \] which vanishes at large distance if only short-range order is present.
By contrast, in a crystal the correlations do not decay away at large separation: the pair correlations retain the periodicity of the underlying lattice. That is the operational meaning of long-range positional order.
1.3.3 Interpretation and Limiting Cases
A few limiting cases are worth keeping in mind.
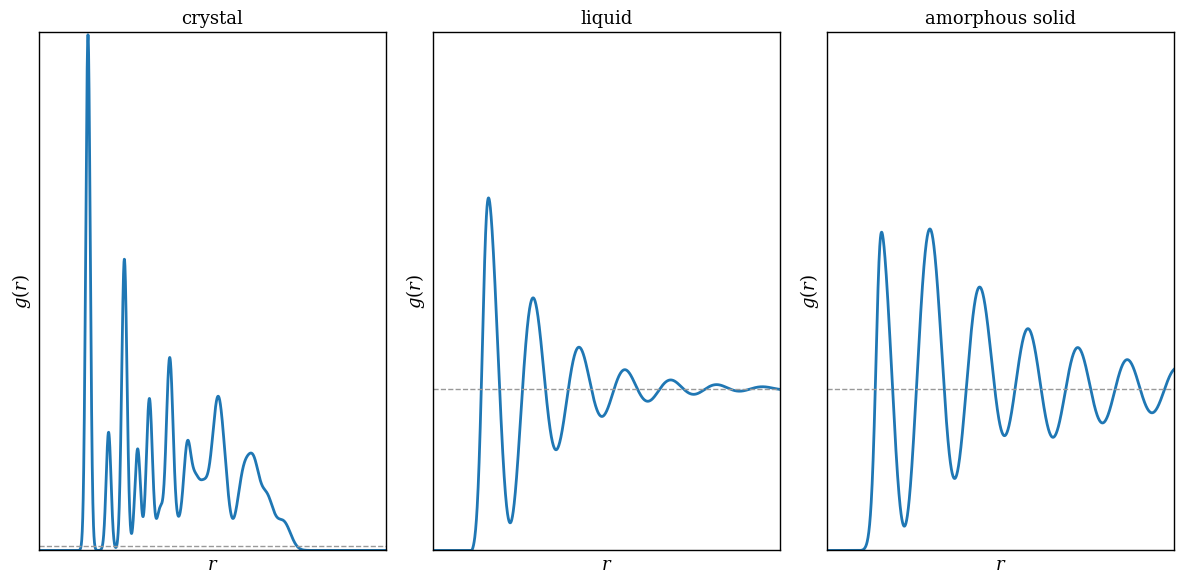
- Completely random arrangement: \(g(r)=1\) at all \(r\), so the presence of one atom tells us nothing about where another one is.
- Amorphous solid or liquid: \(g(r)\) shows a few pronounced peaks at near-neighbor distances, but those oscillations decay and eventually approach \(1\).
- Crystal: correlations remain ordered at arbitrarily large distance; the structure is not described by a few local peaks only.
The pair correlation functions of crystal, liquid, and amorphous solid are schematically illustrated in Figure 1.1.
In isotropic systems, the average number of neighbors in a spherical shell of radius \(r\) and thickness \(dr\) is \[ dN(r)=4\pi \rho g(r) r^2 dr, \] which gives a direct physical meaning to the radial distribution function.
1.3.4 Reciprocal-Space View: the Structure Factor
The same information can be expressed in reciprocal space through the structure factor: \[ S(\mathbf K) = 1+\rho\int [g(\mathbf r)-1]e^{-i\mathbf K\cdot\mathbf r} d\mathbf r . \]
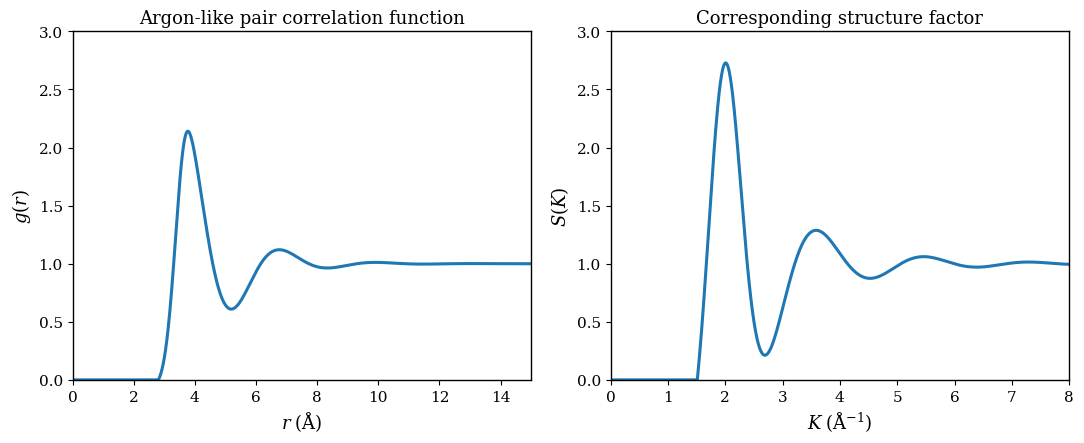
This equation matters because diffraction experiments measure \(S(\mathbf K)\), and from it one infers spatial correlations among atoms.
The liquid-argon example in Figure 1.2 shows very clearly what short-range order looks like in practice. The first-neighbor shell is well defined, but periodic order does not persist indefinitely.
1.4 Structural Types of Solids
1.4.1 Crystalline Solids
A crystalline solid is characterized by long-range positional order. In an ideal crystal, atoms occupy a strictly periodic array, so equivalent atoms have identical neighborhoods no matter how far apart they are.
If the crystallographic axes have the same orientation throughout the sample, the sample is a single crystal.
1.4.2 Polycrystalline Solids
Real materials often consist of many crystalline grains, or crystallites, with different orientations. Such a sample is polycrystalline.
Locally, each grain is crystalline. Globally, the sample no longer has a single orientation. This distinction is important because many bulk thermal measurements average over grain directions and over grain boundaries.
1.4.3 Amorphous Solids
A solid can also be mechanically rigid without having long-range positional order. In an amorphous solid, atoms are frozen in disordered positions. Correlations between distant atoms are lost, but short-range order may still survive.
The essential difference from a liquid is temporal: in an amorphous solid the disordered arrangement is frozen in place on experimental timescales.
1.4.4 Why This Classification Matters for Thermal Physics
This structural classification is not merely descriptive; it determines what kinds of excitations and transport pictures are appropriate.
For thermal physics, the intuition is:
- Crystals support well-defined lattice periodicity, so collective vibrational modes can be labeled by wavevector and branch.
- Polycrystals retain crystalline physics on the scale of a grain, but grain boundaries add scattering and reduce ideal directional behavior.
- Amorphous solids still vibrate and store thermal energy, but the language of perfectly periodic normal modes becomes less exact.
1.5 The Many-Particle Hamiltonian of a Solid
1.5.1 Formal Microscopic Starting Point
The theoretical problem is easy to state and hard to solve. A solid consists of nuclei and electrons interacting through Coulomb forces. In principle, one should solve the many-body Schrödinger equation \[ H\Psi = E\Psi , \] for the full system.
The Hamiltonian is decomposed into nuclear, electronic, and electron–nuclear parts:
which are explicitly defined as
\[ H_{\mathrm{n}} = -\sum_{i=1}^{N_\mathrm{n}}\frac{\hbar^2}{2M_i}\nabla_{\mathbf R_i}^2 + \sum_{i<j}^{N_\mathrm{n}}\frac{Z_i Z_j e^2}{4\pi\varepsilon_0 |\mathbf R_i-\mathbf R_j|}, \]
\[ H_{\mathrm{e}} = -\sum_{i=1}^{N_\mathrm{e}}\frac{\hbar^2}{2m_e}\nabla_{\mathbf r_i}^2 + \sum_{i<j}^{N_\mathrm{e}}\frac{e^2}{4\pi\varepsilon_0 |\mathbf r_i-\mathbf r_j|}, \]
\[ H_{\mathrm{en}} = -\sum_{i=1}^{N_\mathrm{n}}\sum_{j=1}^{N_\mathrm{e}} \frac{Z_i e^2}{4\pi\varepsilon_0 |\mathbf R_i-\mathbf r_j|}. \]
These are the kinetic energies of nuclei and electrons together with the three Coulomb interaction terms.
1.5.2 Why This Problem is Hard
The exact solution is impossible in practice for two separate reasons:
- the number of particles is macroscopically large;
- the Coulomb interaction is strong and long-ranged.
If all interactions were absent, the problem would reduce to independent particles and would be easy. If the number of particles were small, one could still attempt a controlled many-body solution. In a solid, neither of these simplifications holds.
1.5.3 The First Simplification
If the nuclei can be regarded as essentially localized, then their positions define a potential landscape for the electrons, \[ U_{\mathrm{n}}(\mathbf r) = -\sum_{i=1}^{N_\mathrm{n}} \frac{Z_i e^2}{4\pi\varepsilon_0 |\mathbf R_i-\mathbf r|}, \] and one may begin to separate the electronic and ionic aspects of the problem.
This is not yet the Born–Oppenheimer approximation, but it points toward it. Later we will exploit the large mass difference between electrons and nuclei to construct an effective potential-energy surface for atomic motion.
1.6 Thermal Properties
1.6.1 Spectrum and Statistical Mechanics
Once the energy spectrum is known, the methods of statistical physics determine thermal equilibrium properties in principle.
At the most abstract level, thermal physics is built from the chain \[ H \longrightarrow {E_n} \longrightarrow Z=\sum_n e^{-\beta E_n} \longrightarrow F=-k_B T\ln Z , \] where \(T\) denotes temperature, \(Z\) the partition function, \(F\) the free energy, \(k_B\) the Boltzmann constant, and \(\beta=(k_B T)^{-1}\).
For equilibrium thermal observables, this is where the information lives. Heat capacity, for example, is controlled by how the internal energy changes with temperature; thermal expansion is controlled by how the free energy shifts with volume or pressure.
However, this formal statement is not yet useful until we identify the physically relevant excitations in a solid.
1.6.2 Roadmap to Thermal Properties
For the present course, the roadmap is:
This roadmap should be read as a sequence of increasingly specialized approximations.
- Bonding determines which structure is stable.
- The stable structure defines equilibrium atomic positions.
- The curvature of the energy around equilibrium defines the effective stiffness.
- That stiffness determines the normal vibrational modes.
- Quantization of those modes leads to phonons.
- The phonon spectrum produces the dominant lattice contribution to the heat capacity \(C_V\).
- To describe thermal expansion \(\alpha\) and thermal conductivity \(\kappa\), the strictly harmonic picture is not enough.
1.6.3 Outlook on Thermal Properties
A few remarks help orient the rest of the course.
First, a perfectly rigid lattice would have no lattice heat capacity at all. Thermal physics becomes interesting precisely because atoms move.
Second, a purely harmonic crystal already goes a long way: it gives well-defined normal modes and is the correct starting point for the phonon contribution to \(C_V\).
Third, not every thermal observable is of the same kind.
- Heat capacity is primarily an equilibrium counting problem: how many modes are thermally occupied?
- Thermal expansion is already more subtle, because exact harmonicity would not produce thermal expansion.
- Thermal conductivity is subtler still: it is a transport coefficient, so one must understand both carriers and scattering.
1.7 Outlook on the Next Lectures: From Structure to Phonons
The present lecture has intentionally stopped before any detailed derivation of lattice dynamics. The reason is conceptual: before one writes down displacement fields, force constants, and dispersion relations, one should understand what assumptions make those objects meaningful.
The connection to what follows in the course is:
From structure to equilibrium positions: Crystals are special because long-range order allows us to define reference positions for all atoms.
From the many-particle Hamiltonian to an effective ionic problem: We will not solve the full Coulomb problem of interacting electrons and nuclei directly. Instead, we will use controlled approximations to reduce it to nuclear motion in an effective energy landscape.
From bonding to stiffness: The second derivatives of that energy landscape with respect to atomic displacements define the force constants.
From stiffness to vibrations: Small oscillations about equilibrium produce collective normal modes.
From vibrations to thermal properties: After quantization, these modes become phonons. Their spectrum is the microscopic input for the lattice contribution to thermal observables.
- The distinction between short-range and long-range order is a distinction about correlations, not only about visual appearance.
- Crystalline, polycrystalline, and amorphous solids are all mechanically solid, but they are not microscopically equivalent.
- The fundamental microscopic model of a solid is a coupled many-particle quantum problem of nuclei and electrons interacting through Coulomb forces.
- That exact problem is too hard to solve directly because the particle number is huge and the interactions are strong and long-ranged.
- Thermal properties are ultimately tied to the spectrum of accessible excitations and their statistical occupation.
- In solids, lattice vibrations provide the central microscopic route from structure to thermal behavior.
- Heat capacity, thermal expansion, and thermal conductivity do not all require the same level of approximation.
- The next lectures will replace the formal many-particle starting point by a tractable vibrational description.
1.8 Problem Set
Short-range versus long-range order. Explain, in words and with sketches, how the function \(g(r)\) should look for (i) an ideal crystal, (ii) an amorphous solid, and (iii) a completely random arrangement of atoms. What is the corresponding qualitative behavior of \(S(\mathbf K)\) in each case?
From two-particle density to pair-distribution function. Starting from the definition \[ g(\mathbf r_1,\mathbf r_2)=\frac{P(\mathbf r_1,\mathbf r_2)}{\rho(\mathbf r_1)\rho(\mathbf r_2)}, \] show that for a homogeneous isotropic system the expected number of atoms in a spherical shell of radius \(r\) and thickness \(dr\) around a chosen atom is \[ dN(r)=4\pi \rho\,g(r)\,r^2\,dr. \]
Classifying solids. A sample is built from many micron-sized grains, each internally periodic, but the grain orientations are random. Is the sample crystalline, polycrystalline, or amorphous? Which parts of the sample would you expect to scatter heat-carrying excitations most strongly?
Dissecting the Hamiltonian. In the full Hamiltonian of coupled electrons and nuclei \[ H = H_{\mathrm{n}} + H_{\mathrm{e}} + H_{\mathrm{en}}, \] identify which terms describe:
- nuclear motion,
- electron motion,
- interactions among nuclei,
- interactions among electrons,
- interactions between nuclei and electrons, and write out their explicit definitions. Then explain why fixing the nuclei does not yet solve the electronic many-body problem.
Roadmap to thermal observables. For each observable \(C_V\), \(\alpha\), and \(\kappa\), state which additional physical ingredient is essential beyond “a solid is made of atoms”:
- lattice vibrations,
- anharmonicity,
- scattering and transport. Give a one- or two-sentence justification for each choice.